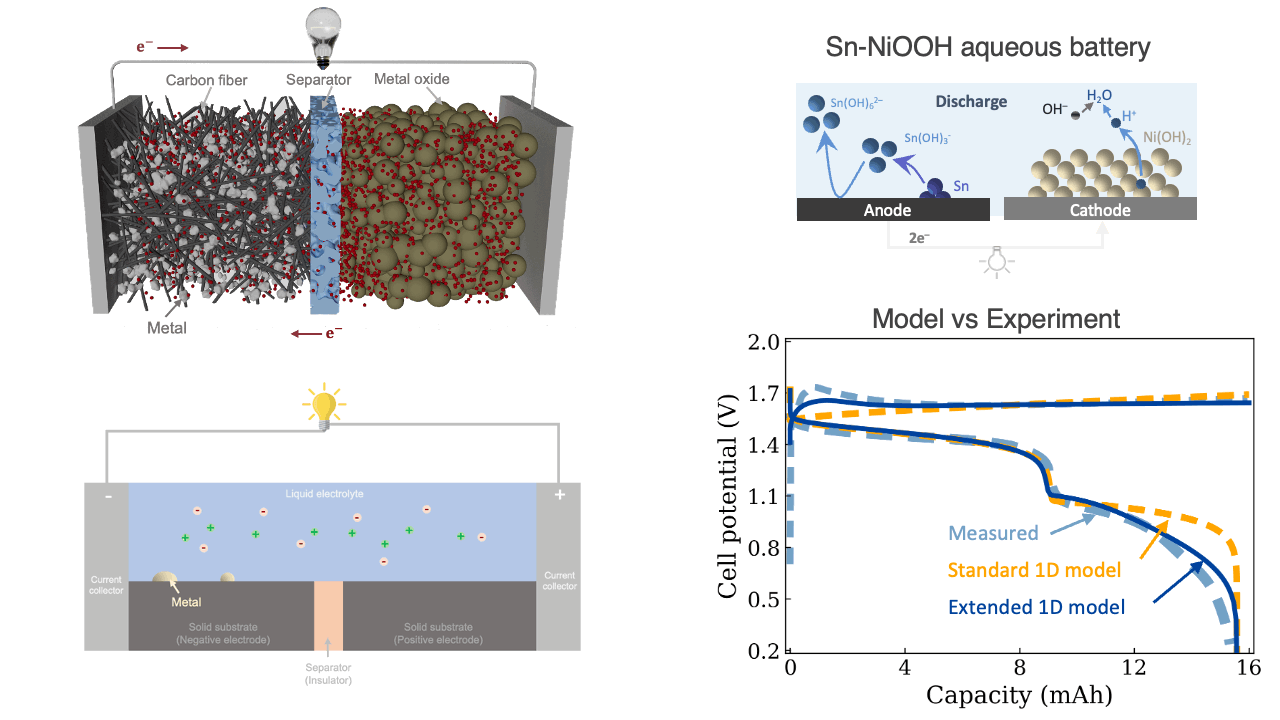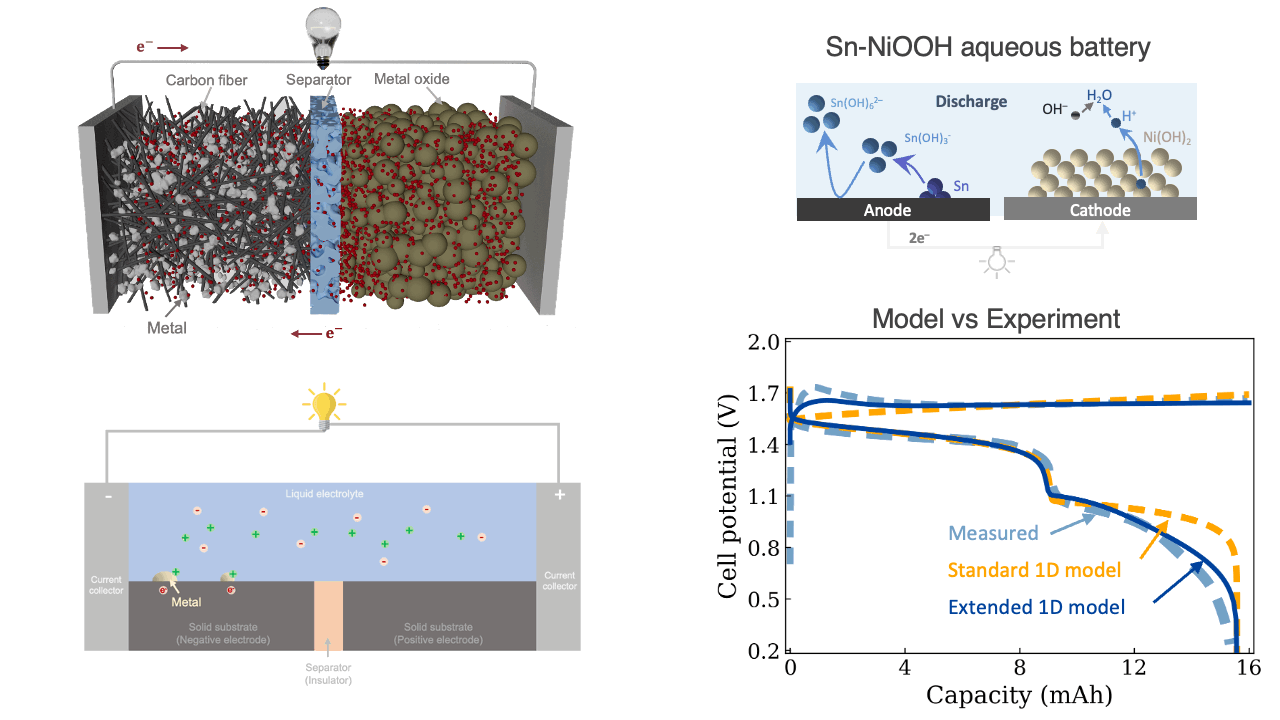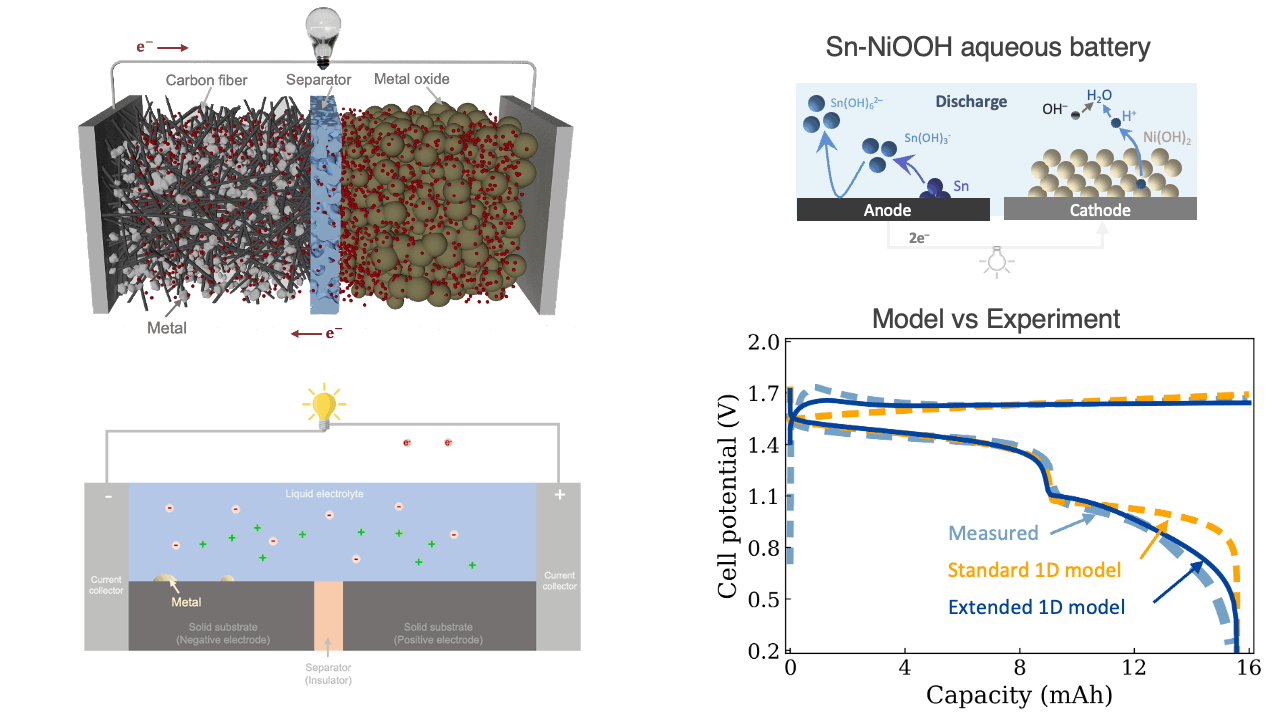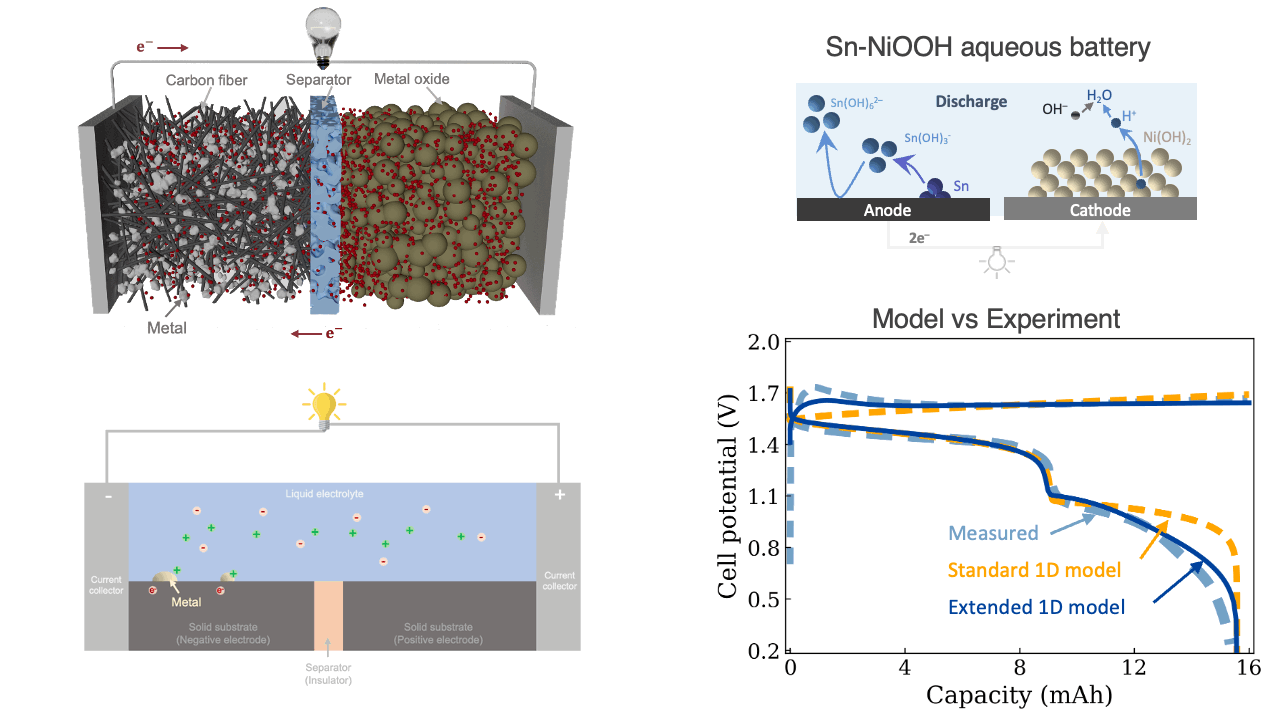
Our paper—entitled “Mathematical Modeling and Experimental Investigations of the Charge–Discharge Mechanisms in Aqueous Batteries“—has been accepted for publication in the Journal of The Electrochemical Society. The paper presents a one-dimensional continuum-scale modeling framework to investigate the reactive transport and charge-transport processes governing aqueous battery charge–discharge behavior. By accounting for multiple chemical and electrochemical reactions—including nucleation, deposition, dissolution, gas evolution, and intercalation—as well as reaction-induced changes in electrolyte pH and reactive surface area, the framework provides a mechanistic tool for studying diverse aqueous battery chemistries. Model–experiment comparisons for acidic H2–Mn, near-neutral Zn–MnO2, and alkaline Sn–NiOOH systems highlight the key processes controlling battery efficiency and performance. Overall, this work helps advance the design of high-utilization, low-cost aqueous batteries for grid-scale energy storage.